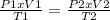Chemistry, 07.10.2019 18:30, dontworry48
Aweather balloon is released into the atmosphere. you know the initial volume, temperature, and air pressure. what information will you need to predict its volume when it reaches its final altitude? which law would you use to calculate this volume?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 21:30, KnMcdonaldk93906
Which substances have the lowest melting points: ionic covalent, or metallic
Answers: 1


Chemistry, 22.06.2019 20:30, dinapaul424
Which states of matter have particles that move independently of one another with very little attraction?
Answers: 1

Chemistry, 23.06.2019 00:00, alisonsolis155
Before it was launched, a helium-filled balloon had a pressure of 201 kpa at a temperature of 27°c. at an altitude of 15,000 m, the pressure had decreased to 2.5 kpa and the temperature had dropped to -14 °c. the volume of the balloon increased to 59.3 m3. what is the original volume of the balloon? 13 m3 0.85 m3 0.077 m3 1.17 m3
Answers: 3
Do you know the correct answer?
Aweather balloon is released into the atmosphere. you know the initial volume, temperature, and air...
Questions in other subjects:


Social Studies, 07.05.2020 05:02

Mathematics, 07.05.2020 05:02



English, 07.05.2020 05:02


Mathematics, 07.05.2020 05:02

History, 07.05.2020 05:02