
Chemistry, 05.10.2019 04:10, shels10tay
Be sure to answer all parts. write the balanced equations corresponding to the following rate expressions: a) rate = − 1 3 δ[ch4] δt = − 1 2 δ[h2o] δt = − δ[co2] δt = 1 4 δ[ch3oh] δt (click in the answer box to activate the palette. do not include states of matter.) b) rate = − 1 2 δ[n2o5] δt = 1 2 δ[n2] δt = 1 5 δ[o2] δt (click in the answer box to activate the palette. do not include states of matter.) c) rate = − 1 2 δ[h2] δt = − 1 2 δ[co2] δt = − δ[o2] δt = 1 2 δ[h2co3] δt (click in the answer box to activate the palette. do not include states of matter.)

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 12:30, azzyla2003
Estructura 7.2 completar repaso 1 - ¿lógico o ilógico? 1 - ¿lógico o ilógico? listen and indicate whether each question and response is lógico or ilógico.
Answers: 3

Chemistry, 22.06.2019 06:00, coolkid2041
One does not belong why? ice, gold ,wood ,diamond and table salt
Answers: 1

Chemistry, 22.06.2019 07:00, shradhwaip2426
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3

Chemistry, 22.06.2019 10:00, nana54muller
Part 1: include important facts found through your research. part 2: include your visual display. include your summary of “the chemistry of water” from the national science foundation website. include your experiment. part 3: include responses to the reflection questions.
Answers: 1
Do you know the correct answer?
Be sure to answer all parts. write the balanced equations corresponding to the following rate expres...
Questions in other subjects:

English, 12.07.2019 14:30


Computers and Technology, 12.07.2019 14:30

Mathematics, 12.07.2019 14:30

Health, 12.07.2019 14:30


History, 12.07.2019 14:30




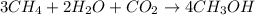
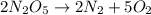
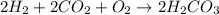
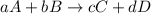
![\text{Rate of disappearance of A}=-\frac{1}{a}\frac{d[A]}{dt}](/tpl/images/0288/1459/2d8eb.png)
![\text{Rate of disappearance of B}=-\frac{1}{b}\frac{d[B]}{dt}](/tpl/images/0288/1459/1e77e.png)
![\text{Rate of formation of C}=+\frac{1}{c}\frac{d[C]}{dt}](/tpl/images/0288/1459/cee4b.png)
![\text{Rate of formation of D}=+\frac{1}{d}\frac{d[D]}{dt}](/tpl/images/0288/1459/7ef32.png)
![Rate=-\frac{1}{a}\frac{d[A]}{dt}=-\frac{1}{b}\frac{d[B]}{dt}=+\frac{1}{c}\frac{d[C]}{dt}=+\frac{1}{d}\frac{d[D]}{dt}](/tpl/images/0288/1459/d4b94.png)
![Rate=-\frac{1}{3}\frac{d[CH_4]}{dt}=-\frac{1}{2}\frac{d[H_2O]}{dt}=-\frac{d[CO_2]}{dt}=+\frac{1}{4}\frac{d[CH_3OH]}{dt}](/tpl/images/0288/1459/a1c90.png)
![Rate=-\frac{1}{2}\frac{d[N_2O_5]}{dt}=+\frac{1}{2}\frac{d[N_2]}{dt}=+\frac{1}{5}\frac{d[O_2]}{dt}](/tpl/images/0288/1459/697db.png)
![Rate=-\frac{1}{2}\frac{d[H_2]}{dt}=-\frac{1}{2}\frac{d[CO_2]}{dt}=-\frac{d[O_2]}{dt}=+\frac{1}{2}\frac{d[H_2CO_3]}{dt}](/tpl/images/0288/1459/13070.png)




