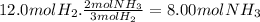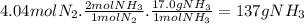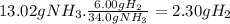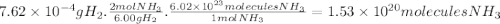How many moles of nh3 can be produced from 12.0 mol of h2 and excess n2? express your answer numerically in moles. view available hint(s) nothing mol nh3m o l n h 3 part b how many grams of nh3 can be produced from 4.04 mol of n2 and excess h2. express your answer numerically in grams. view available hint(s) nothing g nh3g n h 3 part c how many grams of h2 are needed to produce 13.02 g of nh3? express your answer numerically in grams. view available hint(s) nothing g h2g h 2 part d how many molecules (not moles) of nh3 are produced from 7.62×10−4 g of h2? express your answer numerically as the number of molecules. view available hint(s) nothing moleculesm o l e c u l e s

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 09:20, lanaiheart7
How have the greenhouse gasses increased from the year 2000 to 2018
Answers: 2

Chemistry, 23.06.2019 01:00, jaidencoolman2510
Na chemical reaction, activation energy increases the of the reactants. this outcome causes the particles to collide, which results in the of new products.
Answers: 2

Chemistry, 23.06.2019 23:50, SoccerHalo
In the following reaction, oxygen is the excess reactant. sicl4 + o2 → sio2 + cl2 the table shows an experimental record for the above reaction. experimental record trial starting amount of sicl4 starting amount of o2 actual yield of sio2 1 120 g 240 g 38.2 g 2 75 g 50 g 25.2 g part 1: calculate the percentage yield for sio2 for trial 1. also, determine the leftover reactant for the trial. show your work. part 2: based on the percentage yield in trial 2, explain what ratio of reactants is more efficient for the given reaction.
Answers: 3
Do you know the correct answer?
How many moles of nh3 can be produced from 12.0 mol of h2 and excess n2? express your answer numeri...
Questions in other subjects:

Mathematics, 04.04.2020 15:54

Mathematics, 04.04.2020 15:54


History, 04.04.2020 15:54

Mathematics, 04.04.2020 15:54

Spanish, 04.04.2020 15:55

Mathematics, 04.04.2020 15:55