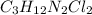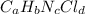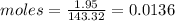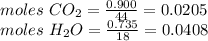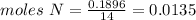Chemistry, 04.10.2019 20:20, estrellagutierrez12
Compound x contains only carbon, hydrogen, nitrogen, and chlorine. when 1.00 g of x is dissolved in water and allowed to react with excess silver nitrate, agno3, all the chlorine in x reacts and 1.95 g of solid agcl is formed. when 1.00 g of x undergoes complete combustion, 0.900 g of co2 and 0.735 g of h2o are formed. what is the empirical formula of x?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:00, misspicafunpoke
Indicate whether the specified alkyl halides will form primarily substitution products, only elimination products, both substitution and elimination products, or no products when they react with sodium methoxide. 1-bromobutane 1-bromo-2-methylpropane 2-bromobutane 2-bromo-2-methylpropane
Answers: 2

Chemistry, 22.06.2019 07:30, nayiiii1874
What three things determine the shape and size of a puddle when water is poured out onto a surface
Answers: 2

Chemistry, 22.06.2019 07:40, sadcase85
22. a flask containing 450 ml of 0.50 m h2so4 was accidentally knocked to the floor. how many grams of nahco, do you need to put on the spill to neutralize the acid according to the following equation: h2so4(aq)+2 nahcos(aq) na, so(aq) +2 h20()+2 co2(g) d) 38 g a) 2.3 g b) 9.5 g c) 19 g
Answers: 1
Do you know the correct answer?
Compound x contains only carbon, hydrogen, nitrogen, and chlorine. when 1.00 g of x is dissolved in...
Questions in other subjects:



Mathematics, 04.01.2021 18:00

Social Studies, 04.01.2021 18:00

Mathematics, 04.01.2021 18:00

Mathematics, 04.01.2021 18:00

Arts, 04.01.2021 18:00

Mathematics, 04.01.2021 18:00

Geography, 04.01.2021 18:00