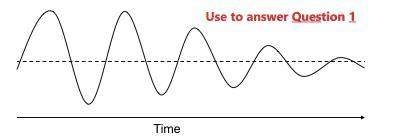
1) as time passes, which of these is decreasing?
a) amplitude
b) crest
c) troug...

Chemistry, 04.10.2019 20:10, karinalovez
1) as time passes, which of these is decreasing?
a) amplitude
b) crest
c) trough
d) wavelength
2)which graph best represents the relative speeds of electromagnetic and mechanical waves through space?
a)
b)
c)
d)
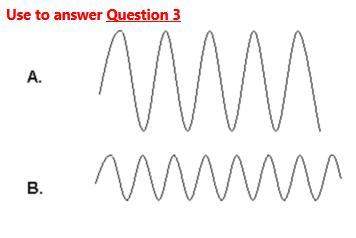
3)which of these waves would have the greatest amount of energy?
a) a
b) b
c) their energy is equal
d) there is not enough information
4)at sunset, the sun appears reddish. what is most likely the reason for this phenomenon?
a) sunlight travels through a longer distance at sunset and interacts with more particles.
eliminate
b) sunlight travels through a shorter distance at sunset and interacts with more particles.
c) sunlight travels through a longer distance at sunset and interacts with fewer particles.
d) sunlight travels through a shorter distance at sunset and interacts with fewer particles.
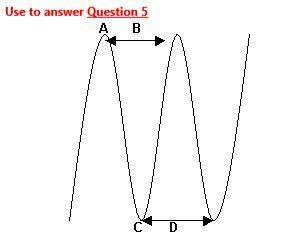
5)the trough of a transverse wave is best shown by choice
a) a.
b) b.
c) c.
d) d.
7)mr. mann's students were studying the properties of light in science class. he told them to put a pencil in a beaker of water, make observations, and then draw what they observed. one group drew the picture seen here. mr. mann explained that when light passes from air into the water, the light waves bend, or and the pencil appears to be broken.
a) diffract
b) reflect
c) refract
d) transact


Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:30, greekfreekisdbz
What type of reaction is shown below? check all that apply. 2h2o2 → 2h2o + o2 synthesis decomposition combustion
Answers: 3


Chemistry, 22.06.2019 09:00, alydiale584
Particles vibrate in a rigid structure and do not move relative to their neighbors.
Answers: 1

Chemistry, 22.06.2019 12:30, pup88
According to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? view available hint(s) according to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? trigonal bipyramidal tetrahedral square planar determination of electron geometry requires information on whether the electron groups are lone pairs or bonding groups.
Answers: 2
Do you know the correct answer?
Questions in other subjects:



Social Studies, 16.09.2021 23:20




Mathematics, 16.09.2021 23:20

Mathematics, 16.09.2021 23:20


Mathematics, 16.09.2021 23:20






