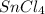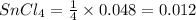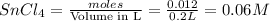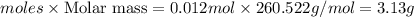Chemistry, 02.10.2019 19:30, domzilla115
Wenzhou prepares 200 ml of a solution of sncl4in which the concentration ofchloride ions is 0.240m. a) what is the molarity of the sncl4solution (i. e. what should the bottle be labeled)? b) what mass of sncl4did wenzhou use?

Answers: 1
Other questions on the subject: Chemistry




Chemistry, 22.06.2019 20:10, maddie1776
Insoluble sulfide compounds are generally black in color. which of the following combinations could yield a black precipitate? check all that apply. na2s(aq)+kcl(aq) li2s(aq)+pb(no3)2(aq) pb(clo3)2(aq)+nano3(aq) agno3(aq)+kcl(aq) k2s(aq)+sn(no3)4(aq)
Answers: 1
Do you know the correct answer?
Wenzhou prepares 200 ml of a solution of sncl4in which the concentration ofchloride ions is 0.240m....
Questions in other subjects:


Mathematics, 20.10.2020 23:01

Mathematics, 20.10.2020 23:01






Mathematics, 20.10.2020 23:01

Mathematics, 20.10.2020 23:01


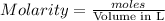
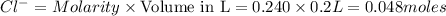
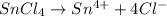
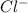 , there is 1 mole of
, there is 1 mole of