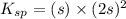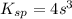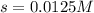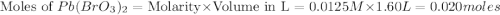Chemistry, 02.10.2019 19:30, ashleyortego5106
Calculate the mass of precipitate that will form if 0.75 l of 0.50 m lead nitrate is mixed with 0.850 l of concentrated sodium bromate. ksp for lead bromate = 7.9 x 10-6.

Answers: 3
Other questions on the subject: Chemistry



Do you know the correct answer?
Calculate the mass of precipitate that will form if 0.75 l of 0.50 m lead nitrate is mixed with 0.85...
Questions in other subjects:






Mathematics, 09.06.2020 20:57



Social Studies, 09.06.2020 20:57

Mathematics, 09.06.2020 20:57


 formed is 164.4 grams.
formed is 164.4 grams.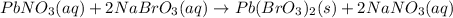
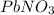 .
.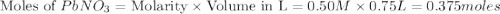

![K_{sp}=[Pb^{2+}][BrO_3^-]^2](/tpl/images/0283/9224/ddb4e.png)