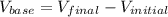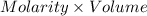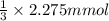Chemistry, 01.10.2019 18:30, SundaeSunday
Cedrick & astrid titrated a 20.00 ml aliquot of grapefruit juice with a 0.165 m naoh solution to the end point. the initial buret reading was 1.72 ml and the final buret reading was 15.51 ml. they calculated that there was 0.1457 g of citric acid present in the juice sample. what is the amount mg of citric acid present per ml of juice?

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 18:30, lattimorekeonna1
How many moles of bromine are needed to produce 3.23 moles of potassium bromide
Answers: 1

Chemistry, 22.06.2019 23:00, lufung8627
Consider the reaction: 2al(s) + fe2o3(s) → al2o3(s) + 2fe(s) the δhf for fe2o3(s) = -824.3 kj/mole. the δhf for al2o3(s) = -1675.7 kj/mole. finish the equation. δhrxn = [(1)( kj/mole) + (2)( kj/mole)] - [(1)( kj/mole) + (2) ( kj/mole)]
Answers: 1

Chemistry, 23.06.2019 01:00, Johnson926
Which elements are found in glucose, the product of photosynthesis? a. carbon, hydrogen, and oxygen b. carbon and hydrogen c. carbon, nitrogen, and oxygen d. hydrogen, nitrogen, and carbon
Answers: 2
Do you know the correct answer?
Cedrick & astrid titrated a 20.00 ml aliquot of grapefruit juice with a 0.165 m naoh solution t...
Questions in other subjects:

Mathematics, 08.07.2019 09:30



Health, 08.07.2019 09:30

History, 08.07.2019 09:30



Mathematics, 08.07.2019 09:30

Mathematics, 08.07.2019 09:30