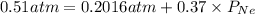Chemistry, 28.09.2019 03:30, dylanclark4965
Agas mixture has a total pressure of 0.51 atm and consists of he and ne. if the partial pressure of he in the mixture is 0.32 atm, what is the partial pressure of the ne in the mixture? enter your answer in the provided box. atm

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 18:00, kingamir
Answer asap need it by wednesday morning carry out the following calculations on ph and ka of from data. i. calculate the ph of 0.02m hcl ii. calculate the ph of 0.036m naoh iii. calculate the ph of 0.36m ca(oh)2 iv. calculate the ph of 0.16m ch3cooh which has ka = 1.74 x 10-5 mol dm-3 v. calculate ka for weak acid ha which has a ph of 3.65 at 0.30m concentration vi. calculate the ka of a solution made by mixing 15.0 cm3 0.2m ha and 60.0 cm3 0.31m a-. [ph= 3.80] vii. calculate the ph of a solution made by mixing 15.0 cm3 0.1m naoh and 35.0 cm3 0.2m hcooh. [ka = 1.82 x 10-4 m]
Answers: 1

Chemistry, 23.06.2019 01:00, davelopez979
An unsaturated hydrocarbon is a hydrogen-carbon compound with a. a network solid structure b. single bonds c. single bonds in a branched-chain structure d. double or triple bonds
Answers: 1
Do you know the correct answer?
Agas mixture has a total pressure of 0.51 atm and consists of he and ne. if the partial pressure of...
Questions in other subjects:

Mathematics, 23.11.2020 23:40

History, 23.11.2020 23:40



Mathematics, 23.11.2020 23:40




Mathematics, 23.11.2020 23:40



 = partial pressure of gas 1
= partial pressure of gas 1 = mole fraction of gas 1
= mole fraction of gas 1 = total pressure of the gases
= total pressure of the gases 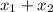 = 1
= 1 


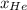 = 0.63
= 0.63 = 1
= 1