

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 03:30, tbeck225
If a solution is considered basic, then a) the hydroxide ion and hydronium ion concentrations are equal. b) the hydroxide ion concentration is less than the hydronium ion concentration. c) the hydronium ion concentration is greater than the hydroxide ion concentration. d) the hydroxide ion concentration is greater than the hydronium ion concentration.
Answers: 1

Chemistry, 22.06.2019 12:30, AnastasiaJauregui
Which of the following describes a compound? (hint: carbon and oxygen bo a. a piece of pure carbon, containing only carbon atoms b. oxygen gas surrounding a solid piece of carbon c. a substance made of two oxygen atoms for each carbon atom carbon and oxygen atoms mixed without being bonded together
Answers: 1
Do you know the correct answer?
Calculate the pressure, in atmospheres, required to compress a sample of helium gas from 27.3 l (at...
Questions in other subjects:


English, 01.09.2019 01:30


Biology, 01.09.2019 01:30



Mathematics, 01.09.2019 01:30


History, 01.09.2019 01:30




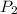 :
:





