
Chemistry, 28.09.2019 03:10, Tonyang1742
Calculate as° for the reaction below: n2(g)+202(g) 2no2(g) where aso for n2(g), o2(g), & no2(g), respectively, is 191.5, 205.0, & 240.5 j/mol-k -156.0 j/k 156.0 j/k 120.5 j/k -120.5 j/k o ooo

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 12:20, missayers172
Achemistry student weighs out 0.306 g of citric acid (h3c6h5o7), a triprotic acid, into a 250 ml volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with 0.1000 m naoh solution. calculate the volume of naoh solution the student will need to add to reach the final equivalence point. be sure your answer has the correct number of significant digits.
Answers: 3

Chemistry, 22.06.2019 16:40, roderickhinton
The diagram below shows the movement of particles. what does this piece of evidence best support? the collision theory the maxwell-boltzmann distribution the effect of pressure on reaction rates the effect of temperature on reaction rates
Answers: 3
Do you know the correct answer?
Calculate as° for the reaction below: n2(g)+202(g) 2no2(g) where aso for n2(g), o2(g), & no2(g...
Questions in other subjects:

Social Studies, 16.07.2019 20:00

Biology, 16.07.2019 20:00


Social Studies, 16.07.2019 20:00

Social Studies, 16.07.2019 20:00


Chemistry, 16.07.2019 20:00

History, 16.07.2019 20:00



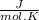 - [ 1 mol × 191,5
- [ 1 mol × 191,5 





