
Chemistry, 27.09.2019 03:30, Knownothing
Ethyl iodide (c2h5i) decomposes at a certain temperature in the gas phase as follows: c2h5i(g) → c2h4(g) + hi(g) from the following data determine the order of the reaction and the rate constant. time (min) [c2h5i] (m) 0.0 2.00 15.0 1.82 30.0 1.64 48.0 1.42 75.0 1.10

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 19:40, trodgers0202
Scientists have developed an explanation of a phenomenon from several verified hypotheses. the explanation has been confirmed through numerous experimental tests. which option best describes this explanation? a. scientific lawb. research questionc. hypothesisd. scientific theory
Answers: 3

Chemistry, 22.06.2019 22:20, trockout4868
How do cfcs cause ozone depletion? how do cfcs cause ozone depletion? ultraviolet radiation breaks down cfcs, molecules containing chlorine. chlorine then breaks one oxygen atom away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation breaks down cfcs, molecules containing chlorine. chlorine then breaks two oxygen atoms away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation creates cfcs, molecules containing chlorine. chlorine then breaks two oxygen atoms away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation creates cfcs, molecules containing chlorine. chlorine then breaks one oxygen atom away from ozone, leaving behind a paired oxygen molecule.
Answers: 2

Chemistry, 23.06.2019 01:30, jonmorton159
At a certain temperature the rate of this reaction is first order in hi with a rate constant of : 0.0632s2hig=h2g+i2g suppose a vessel contains hi at a concentration of 1.28m . calculate how long it takes for the concentration of hi to decrease to 17.0% of its initial value. you may assume no other reaction is important. round your answer to 2 significant digits.
Answers: 1

Do you know the correct answer?
Ethyl iodide (c2h5i) decomposes at a certain temperature in the gas phase as follows: c2h5i(g) → c2...
Questions in other subjects:

Mathematics, 04.02.2020 04:52








History, 04.02.2020 04:52

History, 04.02.2020 04:52


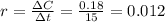
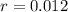 .
.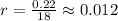
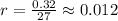
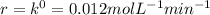 that is, the reaction is the zero order respect to C2H5I since it is not depending on concentration of C2H5I.
that is, the reaction is the zero order respect to C2H5I since it is not depending on concentration of C2H5I. 



