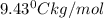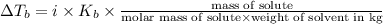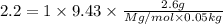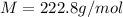A2.60 gram sample of a compound know to contain only indium and chlorine is dissolved in 50.0 g of tin(iv) chloride (kb = 9.43oc kg mol-1). the normal boiling point is raised from 114.1oc for pure sncl4 to 116.3oc for the solution. what is the molecular weight and probable molecular formula of the solute?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:50, ajaydonlee
Select the correct answer how does the heat content of the reaction change in the process of photosynthesis when a glucose molecule is formed? ca the value of is negative the value of qis positive the value of a remains constant the value of a decreases the value of equals zero e
Answers: 2

Chemistry, 22.06.2019 04:40, khan2491
Silver tarnishes as silver metal reacts with hydrogen sulfide, h2s, in the air. in this reaction, dark silver sulfide, au2s, covers the surface of silver. when silver is polished, this coating of silver sulfide can be removed from the surface. this makes the silver shiny again. enter the coefficients that balance the tarnishing reaction equation. (type 1 for no coefficient.)
Answers: 2

Chemistry, 22.06.2019 16:00, hjgjlgkjg
Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chemical reaction takes place in which magnesium combines with oxygen to form magnesium oxide. which of the chemical equations matches the reaction above? a. mg + o2 mgo2 + energy b. 2mg + o mg2o + energy c. 2mg + o2 2mgo + energy d. mg + o mgo + energy
Answers: 1
Do you know the correct answer?
A2.60 gram sample of a compound know to contain only indium and chlorine is dissolved in 50.0 g of t...
Questions in other subjects:

Mathematics, 10.03.2021 16:20

History, 10.03.2021 16:20


Biology, 10.03.2021 16:20

Mathematics, 10.03.2021 16:20



Mathematics, 10.03.2021 16:20

History, 10.03.2021 16:20

Mathematics, 10.03.2021 16:20




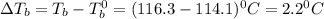 = elevation in boiling point
= elevation in boiling point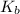 = boiling point constant =
= boiling point constant =