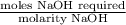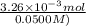Chemistry, 24.09.2019 22:20, PerksInLife
Student weighs out 0.287 g of ascorbic acid (h2ch06), a diprotic acid, into a 250. ml k and dilutes to the mark with distilled water. he plans to titrate the acid with tion olume of naoh ssolution the student will need to add to reach the final equ ur answer to 3 significant digits. ence i don't know submit | privacy oation. all rights reserved. terms of use zo19 micorw

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:20, nyceastcoast
Give the orbital configuration of the phosphorus (p) atom.
Answers: 1


Chemistry, 22.06.2019 21:00, andrethisman88
Kp is the equilibrium constant for dissociation of the propionic acid dimer. what is the sign of the slope for a plot of the natural logarithm of kp vs. inverse temperature for this reaction?
Answers: 1

Chemistry, 23.06.2019 00:30, vane6176
You are attempting to recrystallize a crude product mixture. you add the appropriate amount of hot solvent and are allowing the solution to slowly cool to room temperature. however, at room temperature no crystals have appeared, which of the following methods should be used to induce crystallization? choose all correct answers. a) place the flask in an ice bath. b) swirl the contents of the flask. c) add a small seed crystal of the desired product. d) scratch the inside of the glassware using a stir rod. it can be multiple choices
Answers: 3
Do you know the correct answer?
Student weighs out 0.287 g of ascorbic acid (h2ch06), a diprotic acid, into a 250. ml k and dilutes...
Questions in other subjects:






English, 06.11.2019 08:31


Biology, 06.11.2019 08:31

Geography, 06.11.2019 08:31

English, 06.11.2019 08:31


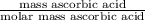
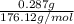
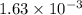 mol
mol
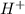 =
= 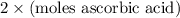
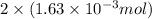
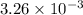 mol
mol