
Asolution contains 0.1 (10-7) moles of hydroxyl ions (oh-) per liter. which of the following best describes this solution? a solution contains 0.1 (10-7) moles of hydroxyl ions (oh-) per liter. which of the following best describes this solution? basic: h+ donor basic: h+ acceptor acidic: h+ acceptor neutral acidic: h+ donor

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:50, sgslayerkingminecraf
Which of the following statements about acidic water is true? a. acid has no effect on the h, o molecules. b. the solution contains a larger number of oh ions than h, o ions. c. the solution contains a larger number of h, o ions than qh ions. d. the solution contains an equal number of h, o ions and oh ions. none of the above e.
Answers: 1

Chemistry, 23.06.2019 00:00, savyblue1724707
How many moles of water are created if 3 moles of hydrogen react completely with excess oxygen?
Answers: 3


Chemistry, 23.06.2019 08:10, andrewrangel63
An experiment is conducted to see if cats preferred skim milk or 2% milk. a cup of skim milkwas put out for 5 kittens and then measured how much the kittens drank over the course of aday. following a cup of 2% milk was purout for the skittens and then masured how much thekittens drank over the course of a day. the same kittens were used and the milk was served atthe same temperature. it was discovered that the cats liked the 2% milk more than the skimmilk. what is the dependent variable in this experiment?
Answers: 1
Do you know the correct answer?
Asolution contains 0.1 (10-7) moles of hydroxyl ions (oh-) per liter. which of the following best de...
Questions in other subjects:

History, 11.12.2020 01:40

Mathematics, 11.12.2020 01:40






English, 11.12.2020 01:40



![pH=-\log [H^+]](/tpl/images/0241/8360/37e81.png)
![pOH=-\log[OH^-]](/tpl/images/0241/8360/fe336.png)
![pOH=-\log[10^{-7}]](/tpl/images/0241/8360/492ed.png)
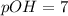
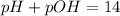
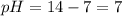
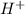 nor donates
nor donates 



