
Chemistry, 14.09.2019 08:30, webbjalia04
In a study of the conversion of methane to other fuels, a chemical engineer mixes gaseous methane and gaseous water in a 0.379 l flask at 1191 k. at equilibrium, the flask contains 0.145 mol of co gas, 0.218 mol of h2 gas, and 0.25 mol of methane. what is the water concentration at equilibrium (kc = 0.30 for this process at 1191 k)?
enter to 4 decimal places.
hint: look at sample problem 17.7 in the 8th ed silberberg book. write a balanced chemical equation. write the kc expression. calculate the equilibrium concentrations of all the species given (moles/liter). put values into kc expression, solve for the unknown.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:30, alexusnicole817
Which of the following signs of a chemical reaction are observed in the reaction of potassium with water? precipitate formed temperature change smell produced gas produced color change
Answers: 2

Chemistry, 22.06.2019 12:10, yootmytoot
Achemistry student needs to standardize a fresh solution of sodium hydroxide. he carefully weighs out of oxalic acid , a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in of distilled water. the student then titrates the oxalic acid solution with his sodium hydroxide solution. when the titration reaches the equivalence point, the student finds he has used of sodium hydroxide solution. calculate the molarity of the student's sodium hydroxide solution. be sure your answer has the correct number of significant digits.
Answers: 1

Chemistry, 22.06.2019 15:20, munziruddin204
Which description best characterizes the motion of particles in a solid?
Answers: 2
Do you know the correct answer?
In a study of the conversion of methane to other fuels, a chemical engineer mixes gaseous methane an...
Questions in other subjects:






Chemistry, 15.08.2020 20:01

Mathematics, 15.08.2020 20:01




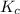
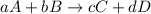
![K_{c}=\frac{[C]^c[D]^d}{[A]^a[B]^b}](/tpl/images/0231/1245/b6f47.png)
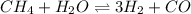
![K_{c}=\frac{[H_2]^3[CO]}{[CH_4][H_2O]}](/tpl/images/0231/1245/e7210.png) ....(1)
....(1)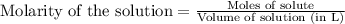
![[CO]=\frac{0.145mol}{0.379L}=0.383mol/L](/tpl/images/0231/1245/315b1.png)
![[H_2]=\frac{0.218mol}{0.379L}=0.575mol/L](/tpl/images/0231/1245/b555b.png)
![[CH_4]=\frac{0.25mol}{0.379L}=0.660mol/L](/tpl/images/0231/1245/35995.png)

![0.30=\frac{(0.575)^3\times 0.383}{0.660\times [H_2O]}](/tpl/images/0231/1245/8e17e.png)
![[H_2O]=\frac{(0.575)^3\times 0.383}{0.660\times 0.30}=0.3677](/tpl/images/0231/1245/f4553.png)





