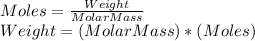Chemistry, 13.09.2019 21:30, ladybuggirl400
Amixture of gases is analyzed and found to have the following volume composition: co2 12.0%, co 6.0, ch4 27.3, h2 9.9, and n2 44.8. how much will 38 [mol] of this mixture weigh, in [kg]?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 09:40, kolibeilfuss
Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid synthesis. an industrial chemist studying this reaction fills a 25.0l tank with 4.5 mol of sulfur dioxide gas and 4.5 mol of oxygen gas at 30.°c. he then raises the temperature, and when the mixture has come to equilibrium measures the amount of sulfur trioxide gas to be 1.4 mol. calculate the concentration equilibrium constant for the reaction of sulfur dioxide and oxygen at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 3

Chemistry, 22.06.2019 10:30, cheyennecarrillo14
If you add 5.00 ml of 0.100 m sodium hydroxide to 50.0 ml of acetate buffer that is 0.100 m in both acetic acid and sodium acetate, what is the ph of the resulting solution? acetic acid: ka = 1.8. x 10-5
Answers: 1


Chemistry, 22.06.2019 14:30, belindajolete
How do temperature and salinity affect deepwater currents? as temperatures and salinity levels of water increase, the water rises to the surface where it creates currents as it moves to colder regions. they create changes in wind direction, moving denser water in the same direction as the wind and causing the deepwater circulation patterns found in the ocean. they equalize the forces on undersea currents caused by the coriolis effect as they replace more dense water with less dense water. they create density differences that cause dense deepwater currents to flow toward the equator where they displace less dense, warmer water above them.
Answers: 2
Do you know the correct answer?
Amixture of gases is analyzed and found to have the following volume composition: co2 12.0%, co 6.0...
Questions in other subjects:


Chemistry, 25.11.2021 05:40

Computers and Technology, 25.11.2021 05:40

Mathematics, 25.11.2021 05:40




Mathematics, 25.11.2021 05:40

History, 25.11.2021 05:40