
Chemistry, 12.09.2019 21:20, seaotter7140
As a technician in a large pharmaceutical research firm, you need to produce 350. ml of 1.00 m potassium phosphate buffer solution of ph = 7.07. the pka of h2po4− is 7.21. you have the following supplies: 2.00 l of 1.00 m kh2po4 stock solution, 1.50 l of 1.00 m k2hpo4 stock solution, and a carboy of pure distilled h2o. how much 1.00 m kh2po4 will you need to make this solution?

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 00:20, paulawells11
What are the spectator ions in 2h+ + so42- + ca2+ + 2r → caso4 + 2h+ + 21?
Answers: 1

Chemistry, 22.06.2019 02:00, webbhlharryteach
In which of these cases are the two wave points considered to be in phase with each other?
Answers: 1

Do you know the correct answer?
As a technician in a large pharmaceutical research firm, you need to produce 350. ml of 1.00 m potas...
Questions in other subjects:


Mathematics, 12.02.2021 22:30

History, 12.02.2021 22:30




Mathematics, 12.02.2021 22:30

Mathematics, 12.02.2021 22:30

Mathematics, 12.02.2021 22:30

Mathematics, 12.02.2021 22:30


![\frac{[HPO4^{2-}] }{[H2PO4^{-}]}](/tpl/images/0229/1588/94f69.png)
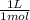 = 0,203 L ≡ 203 mL of 1,00M KH₂PO₄
= 0,203 L ≡ 203 mL of 1,00M KH₂PO₄



