Chemistry, 08.09.2019 23:20, cogger9348
Explain each of the following in terms of dalton's atomic theory.
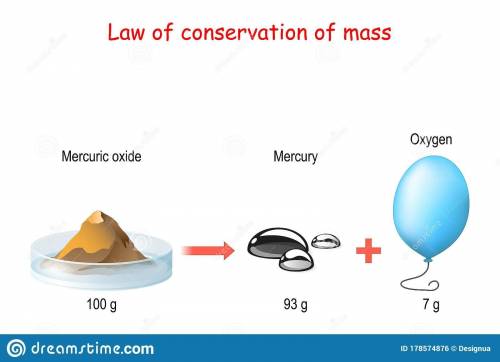
a. the law of conservation of mass
b. the law of definite proportions
c. the law of multiple proportions

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:30, jadepotts3965
Calculate the change in entropy if br2(l) is converted into gaseous br atoms. s° for br2(l) = 152.2 j/(mol•k) s° for br2(g) = 245.5 j/(mol•k) s° for br(g) = 175.0 j/(mol•k)
Answers: 2

Chemistry, 21.06.2019 20:30, sandersmakaylaovq5vu
The balanced chemical equation for this lab is: 3cucl2(aq) + 2al(s) 3cu(s) + 2alcl3(aq) if 10.5 g copper chloride react with 12.4 g aluminum, what is the limiting reactant?
Answers: 3


Chemistry, 22.06.2019 17:00, marsjupiter2554
The atoms of a solid aluminum can are close together, vibrating in a rigid structure. if the can is warmed up on a hot plate, what happens to the atoms?
Answers: 1
Do you know the correct answer?
Explain each of the following in terms of dalton's atomic theory.
a. the law of conservation o...
a. the law of conservation o...
Questions in other subjects:


Mathematics, 15.12.2020 20:40



History, 15.12.2020 20:40

Physics, 15.12.2020 20:40


Mathematics, 15.12.2020 20:40

Mathematics, 15.12.2020 20:40