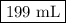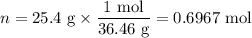Chemistry, 06.09.2019 23:30, kloekamakeeaina14
Muriatic acid, an industrial grade of concentrated hcl, is used to clean masonry and cement. its concentration is 11.7 m. for routine use, a diluted solution of 3.50 m is prepared. how many milliliters of 3.50 m muriatic acid solution contain 25.4 g of hcl?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:30, genyjoannerubiera
Identify two types of chemical bonding in the source of dietary potassium
Answers: 3

Chemistry, 22.06.2019 22:10, zwbaby3693
Which aqueous solution of ki freezes at the lowest temperature? 1) 1 mol of ki in 500. g of water 2) 2 mol of ki in 500. g of water 3) 1 mol of ki in 1000. g of water 4) 2 mol of ki in 1000. g of water
Answers: 3

Chemistry, 23.06.2019 01:30, sheldonwaid4278
Magnesium is the limiting reactant in this experiment. calculate the theoretical yield of mgo for each trial. trial 1: trial 2: data mass of empty crucible with lid trial 1: 26.688 trial 2: 26.681 mass of mg metal, crucible, and lid trial 1: 26.994 trial: 2 26.985 mass of mgo, crucible, and lid trial 1: 27.188 trial 2: 27.180
Answers: 1
Do you know the correct answer?
Muriatic acid, an industrial grade of concentrated hcl, is used to clean masonry and cement. its con...
Questions in other subjects:



Mathematics, 31.07.2021 04:00





World Languages, 31.07.2021 04:00

English, 31.07.2021 04:00

Mathematics, 31.07.2021 04:00