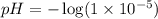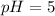Acid & base calculations calculate the hydronium ion concentration for each. tell whether it is an acid or a base. 1. ph = 5.54 2. poh = 9.7 3. ph = 7.0 4. ph = 12.9 5. poh = 1.2 calculate the poh for each. tell whether it is an acid or a base. 6. [h3o+] = 1 x 10-5 m

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:30, josephaciaful
Follow the steps provided in the simulation to add water to the graduated cylinder, select one of the three samples (copper, silver, or gold), set its mass to the values given in the statements below, and calculate its density. here is a summary of the steps required: add water by clicking and holding prepare a known volume of water button. until the desired volume of water has been added. if more than the desired volume is added, click the reset button. button and redo the procedure. a single click will add about 21.0 ml of water. to set the mass, click and hold weigh out metal button. until the desired amount of metal is added to the weighing pan. once the desired mass of the metal is added, release the button. transfer the metal to water and then click on calculate density button. to see how the density is calculated using water displacement to measure the volume of the solid. to save time you can approximate the initial volume of water to â±1 ml and the initial mass of the solid to â±1 g. for example, if you are asked to add 23 ml of water, add between 22 ml and 24 ml. which metals in each of the following sets will have equal density? check all that apply.
Answers: 1

Chemistry, 22.06.2019 05:00, pandasarecute53
If the density of water is 1.0 g/cm3, which of these materials would float in water, based on their densities? check all that apply. aluminum cork iron lead wax
Answers: 1

Chemistry, 22.06.2019 21:50, BookandScienceNerd
Answer the questions about this reaction: nai(aq) + cl2(g) → nacl(aq) + i2(g) write the oxidation and reduction half-reactions: oxidation half-reaction: reduction half-reaction: based on the table of relative strengths of oxidizing and reducing agents (b-18), would these reactants form these products? write the balanced equation: answer options: a. 0/na -> +1/na+1e- b. nai(aq) + cl2(g) → nacl(aq) + i2(g) c. +1/na+1e- -> 0 /na d. -1/2i -> 0/i2+2e- e. no f. 4nai(aq) + cl2(g) → 4nacl(aq) + i2(g) g. 2nai(aq) + cl2(g) → 2nacl(aq) + i2(g) h. 4nai(aq) + 2cl2(g) → 4nacl(aq) + 2i2(g) i. nai(aq) + cl2(g) → nacl(aq) + i2(g) j. 0/cl2+2e -> -1/2cl- k. yes
Answers: 1

Chemistry, 23.06.2019 02:00, matthewsorrow02
What is the mass of 0.750 mole of aluminum oxide, al2o3?
Answers: 1
Do you know the correct answer?
Acid & base calculations calculate the hydronium ion concentration for each. tell whether it is...
Questions in other subjects:


Mathematics, 08.04.2020 01:33

History, 08.04.2020 01:33

Biology, 08.04.2020 01:33


Mathematics, 08.04.2020 01:33


Mathematics, 08.04.2020 01:33



![pH=-\log[H_3O^+]](/tpl/images/0224/7714/a23b5.png) ......(1)
......(1)![5.54=-\log[H_3O^+]](/tpl/images/0224/7714/46ae8.png)
![[H_3O^+]=2.88\times 10^{-6}M](/tpl/images/0224/7714/94865.png)
![4.3=-\log[H_3O^+]](/tpl/images/0224/7714/cd75a.png)
![[H_3O^+]=5.012\times 10^{-5}M](/tpl/images/0224/7714/9980d.png)
![7.0=-\log[H_3O^+]](/tpl/images/0224/7714/bd612.png)
![[H_3O^+]=1.00\times 10^{-7}M](/tpl/images/0224/7714/e9a37.png)
![12.9=-\log[H_3O^+]](/tpl/images/0224/7714/250d5.png)
![[H_3O^+]=1.26\times 10^{-13}M](/tpl/images/0224/7714/8cabb.png)
![12.8=-\log[H_3O^+]](/tpl/images/0224/7714/d6d68.png)
![[H_3O^+]=1.58\times 10^{-13}M](/tpl/images/0224/7714/c23c1.png)
![[H_3O^+]=1\times 10^{-5}M](/tpl/images/0224/7714/3d88c.png)