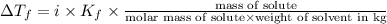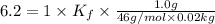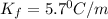Chemistry, 06.09.2019 03:30, ineedtopeebeforethec
Pure nitrobenzene freezes at 5.67 c. when 1.0g of ethanol (c2h6o) is mixed with 20.0g nitrobenzene, the freeze point drops to –0.53 c. what is the freezing-point depression constant (kf) of nitrobenzene?

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 07:30, reaperqueen21
Compare and contrast the bohr model and the electron cloud models of the atom.
Answers: 1

Chemistry, 22.06.2019 14:00, hannahhoskings6989
What was bohr’s contribution to the planetary model
Answers: 1
Do you know the correct answer?
Pure nitrobenzene freezes at 5.67 c. when 1.0g of ethanol (c2h6o) is mixed with 20.0g nitrobenzene,...
Questions in other subjects:





Social Studies, 06.04.2021 18:00






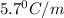
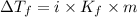
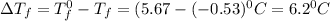 = Depression in freezing point
= Depression in freezing point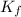 = freezing point constant = ?
= freezing point constant = ?