
Chemistry, 04.09.2019 02:20, jeanetteelliotp9ru6m
Consider two gases, a and b, each in a 1.0-l container with both gases at the same temperature and pressure. the mass of gas a in the container is 0.34 g and the mass of gas b in the container is 0.48 g.
a. which gas sample has the most molecules present? b. which gas sample has the largest average kinetic energy? c. which gas sample has the fastest average velocity? d. how can the pressure in the 2 containers be equal to each other since the larger gas b molecules collide with the container walls more forcefully?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:20, jtingley0502
Temperature is _related to the average kinetic energy of a gas. inversely directly not disproportionally
Answers: 1


Chemistry, 22.06.2019 15:00, NatalieKnows
Areaction is first order with respect to reactant x and second order with respect to reactant y. which statement describes the rate law for this reaction?
Answers: 1
Do you know the correct answer?
Consider two gases, a and b, each in a 1.0-l container with both gases at the same temperature and p...
Questions in other subjects:

Computers and Technology, 20.08.2019 03:30



Social Studies, 20.08.2019 03:30

Social Studies, 20.08.2019 03:30


Business, 20.08.2019 03:30

History, 20.08.2019 03:30


Mathematics, 20.08.2019 03:30


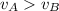 D) compensation between force and velocity.
D) compensation between force and velocity.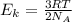 , where R is the ideal gas constant, T is the temperature and
, where R is the ideal gas constant, T is the temperature and 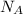 is the Avogadro´s number. All this values are the same in both gases thus they have the same kinetic energy.
is the Avogadro´s number. All this values are the same in both gases thus they have the same kinetic energy.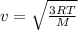 , where M is the molecular mass, if we want to know wich gas has fastest velocity is enough to know the rate between two velocities.
, where M is the molecular mass, if we want to know wich gas has fastest velocity is enough to know the rate between two velocities.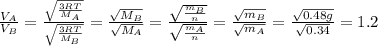 , due to rate is greater than 1
, due to rate is greater than 1 



