
Propane c3h8 is a hydrocarbon tht is commonly used as a fuel
(a)write a balanced equation for the complete combustion used propane gas.
(b) calculate the volume of air at 25c and 1.00 atmosphere that is needed to completely combust 25.0 grams of propna. assume that air is 21.0 percnt o2 by volume. (hint: we will see how to do this calcullation in a later chapter on gases -- for now use the infomation that 1.00l of air at 25c and 1.00atm contains 0.275 g of o2 pe liter.)
(c) the heat of combustion of propane is -2,219.2 kj/mol. calculate the heat of formation, hof of propane given that hof of h2o = -285.8 kj/mol and hof of co2 = -393.5 kj/mol
(d) assuming that all of the het releasd in burning 25.0 grams of propane is transferred to 4.00 kilograms of water calculate the increase in temperature of water .

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:30, coryoddoc3685
Turbo the snail moves across the ground at a pace of 12 feet per day. if the garden is 48 feet away, how many days will it take for the snail to get there?
Answers: 2

Chemistry, 22.06.2019 11:00, hannah5143
The human eye contains a molecule called 11-cis-retinal that changes shape when struck with light of sufficient energy. the change in shape triggers a series of events that results in an electrical signal being sent to the brain that results in vision. the minimum energy required to change the conformation of 11-cis-retinal within the eye is about 164 kj/mol.
Answers: 2

Chemistry, 22.06.2019 14:00, hammackkatelyn60
The content of manganese (mn) in steel was determined spectrophotometrically and with the use of the standard addition method. an unknown sample of mn from a digested steel sample gave an absorbance of 0.185 when analyzed spectrophotometrically. when 5.00 ml of solution containing 95.5 ppm mn was added to 50.0 ml of the unknown steel solution (digested sample), the absorbance was 0.248. calculate the concentration, in parts-per-million (ppm), of mn in the digested steel sample solution.
Answers: 3
Do you know the correct answer?
Propane c3h8 is a hydrocarbon tht is commonly used as a fuel
(a)write a balanced equation for...
(a)write a balanced equation for...
Questions in other subjects:

Biology, 12.10.2019 12:50



Social Studies, 12.10.2019 12:50


History, 12.10.2019 12:50

Mathematics, 12.10.2019 12:50

History, 12.10.2019 12:50

Mathematics, 12.10.2019 12:50

Biology, 12.10.2019 12:50


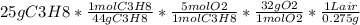
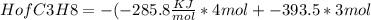
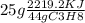 = 1260.9KJ
= 1260.9KJ



