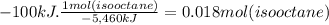Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 15:30, lovebaeforlife351
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. predict the effect (shift right, shift left, or no effect) of the indicated volume change. drag the appropriate items to their respective bins. co(g) + h2o(g) < => co2(g) + h2(g) (volume is decreased) pcl3(g) + cl2(g) < => pcl5(g) (volume is increased) caco3(s)< => cao(s) + co2(g) (volume is increased)
Answers: 1

Chemistry, 22.06.2019 16:30, joshua1255
Find the number of moles of argon in 364g of argon.
Answers: 2
Do you know the correct answer?
How many moles of isooctane must be burned to produce 100 kj of heat under standard state condaition...
Questions in other subjects:

Biology, 16.11.2020 01:00




Business, 16.11.2020 01:00



English, 16.11.2020 01:00

Business, 16.11.2020 01:00