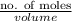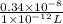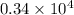Chemistry, 02.09.2019 19:30, elijahjacksonrp6z2o7
The level of mercury in a stream was suspected to be above the minimum considered safe (1part per million by weight). an analysis indicated that the concentration was 0.68 parts per million .assume a density of 1.0 g/ml and calculate the molarity of mercury is the stream.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:00, paynedeforest2596
According to the tide table below what time of day will the highest tide occur? (2 pt) the highest tide will occur at
Answers: 1

Chemistry, 22.06.2019 12:10, purplefish53
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 12:20, missayers172
Achemistry student weighs out 0.306 g of citric acid (h3c6h5o7), a triprotic acid, into a 250 ml volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with 0.1000 m naoh solution. calculate the volume of naoh solution the student will need to add to reach the final equivalence point. be sure your answer has the correct number of significant digits.
Answers: 3

Chemistry, 22.06.2019 14:30, Cartucho1978
According to le chatelier’s principle, a system in chemical equilibrium responds to stress by shifting the equilibrium in a direction that reduces the stress. normalizes the stress. increases the stress. changes the stress.
Answers: 1
Do you know the correct answer?
The level of mercury in a stream was suspected to be above the minimum considered safe (1part per mi...
Questions in other subjects:

Chemistry, 07.07.2019 09:30


History, 07.07.2019 09:30




Mathematics, 07.07.2019 09:30

History, 07.07.2019 09:30


Social Studies, 07.07.2019 09:30


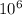 mg of solution.
mg of solution.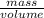
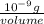
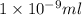 (as 1 mg = 0.001 g)
(as 1 mg = 0.001 g)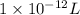 .
.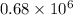 g
g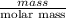
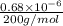
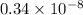 mol
mol