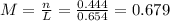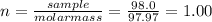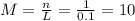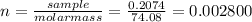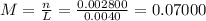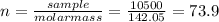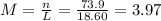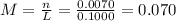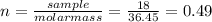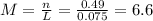Chemistry, 02.09.2019 18:20, nyraimccall408
Determine the molarity for each of the following solution solutions:
(a)0.444 mol of cocl2 in 0.654 l of solution
(b) 98.0 gram of phosphoric acid, h3po4, in 100 l of solution
(c) 0.2074 g of calcium hydroxide, ca(oh)2 in 40.00 ml of solution
(d)10.5 kg of na2ao4.10h2o in 18.60 l of solution
(e) 7.0 x 10-3 mol of l2 in 100.0 ml of solution
(f) 1.8 x 104 mg of hcl in 0.075 of solution

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:30, acaciacoats
The atomic radius of sodium is 186 pm and of chlorine is 100 pm. the ionic radius for na+ is 102 pm and for cl– is 181 pm. in going from na to cl in period 3, why does the atomic radius decrease while the ionic radius increases? a. the inner electrons in the sodium cation shield its valence electrons more effectively than the inner electrons in the chloride anion do. b. the inner electrons shield the valence electrons more effectively in the chlorine atom than in the chloride anion. c. the outermost electrons in chloride experience a smaller effective nuclear charge than those in the sodium cation do. d. the outermost electrons in chloride experience a larger effective nuclear charge than those in the sodium cation do. e. monatomic ions are bigger than the atoms from which they are formed.
Answers: 2


Chemistry, 22.06.2019 10:00, micahwilkerson9495
Select all of the methods through which a drug can enter your body. injection swallowing inhalation absorption
Answers: 2
Do you know the correct answer?
Determine the molarity for each of the following solution solutions:
(a)0.444 mol of cocl2 i...
(a)0.444 mol of cocl2 i...
Questions in other subjects:

History, 19.10.2019 10:30

Business, 19.10.2019 10:30

Biology, 19.10.2019 10:30



Social Studies, 19.10.2019 10:30

Mathematics, 19.10.2019 10:30


Mathematics, 19.10.2019 10:30