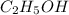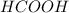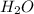Chemistry, 02.09.2019 16:10, kyleejokow
Which contains the greatest mass of oxygen: 0.75 mol of ethanol (c2h5oh), 0.60 mol of formic acid (hco2h) or 1.0 mol
of water (h2o) ? explain why.

Answers: 2
Other questions on the subject: Chemistry




Chemistry, 22.06.2019 13:50, amandamac7339
Abeaker with 2.00×102 ml of an acetic acid buffer with a ph of 5.000 is sitting on a benchtop. the total molarity of acid and conjugate base in this buffer is 0.100 m. a student adds 4.70 ml of a 0.360 m hcl solution to the beaker. how much will the ph change? the pka of acetic acid is 4.740.
Answers: 1
Do you know the correct answer?
Which contains the greatest mass of oxygen: 0.75 mol of ethanol (c2h5oh), 0.60 mol of formic acid (...
Questions in other subjects:




Physics, 28.10.2020 23:00

English, 28.10.2020 23:00


Mathematics, 28.10.2020 23:00

History, 28.10.2020 23:00

Mathematics, 28.10.2020 23:00

Mathematics, 28.10.2020 23:00