
Chemistry, 01.09.2019 00:30, gachaperson123
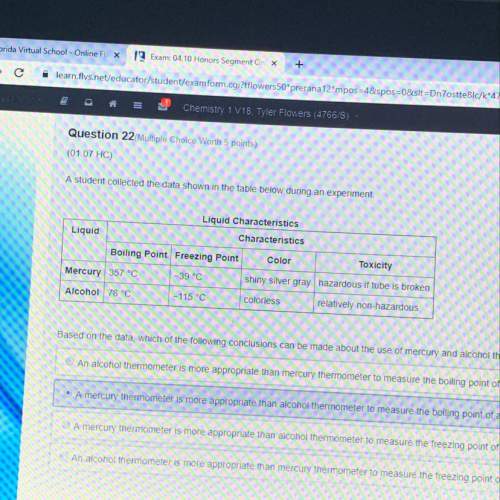
Astudent collected data shown in the table below during an experiment. based on the data, which of the following conclusions can be made about the use of mercury and alcohol thermometers
a) an alcohol thermometer is more appropriate than mercury thermometer to measure the boiling point of a colorless liquid that boils at 100°c
b) a mercury thermometer is more appropriate than an alcohol thermometer to measure the boiling point of a colorless liquid that boils at 82°c
c) a mercury thermometer is more appropriate than alcohol thermometer to measure the freezing point of a colorless liquid that freezes at -40°c
d) an alcohol thermometer is more appropriate than mercury thermometer to measure the freezing point of a colorless liquid that freezes at -20°c

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 23.06.2019 00:00, savyblue1724707
How many moles of water are created if 3 moles of hydrogen react completely with excess oxygen?
Answers: 3


Chemistry, 23.06.2019 08:00, yddlex
Suppose a pair of chemical compounds a and b can react in two different ways: a + b -> c reaction 1 gives product c. a + b -> d reaction 2 gives product d. the following facts are known about the two reactions: . reaction 1 is endothermic and reaction 2 is exothermic. if a reaction vessel is charged (filled) with a and b , then at first d is produced faster than c. use these facts to sketch a qualitative reaction energy diagram for both reactions. note: because these sketches are only qualitative, the energies don? t have to be exact. they only have to have the right relationship to each other. for example, if one energy is less than another, that fact should be clear in your sketch.
Answers: 3
Do you know the correct answer?
Astudent collected data shown in the table below during an experiment. based on the data, which of t...
Questions in other subjects:

Physics, 18.03.2021 02:20


English, 18.03.2021 02:20


Mathematics, 18.03.2021 02:20





Mathematics, 18.03.2021 02:20






