

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 11:00, coco8560
Freezing and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1


Chemistry, 22.06.2019 17:30, nijanicole164
A650 ml sodium bromine solution has a bromide ion concentration of 0.245 m. what is the mass (g) of sodium bromide in solution? a) 103.b)0.00155.c)16400.d) 16.4.e) 0.159
Answers: 2
Do you know the correct answer?
Determine how many atoms of pure silver will be created when 19.83 x 1023 atoms of copper are used i...
Questions in other subjects:



History, 07.05.2020 01:12



History, 07.05.2020 01:12




History, 07.05.2020 01:13


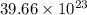
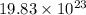
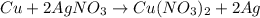
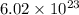 number of copper atoms react to give
number of copper atoms react to give 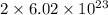 number of silver atoms.
number of silver atoms.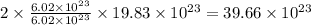 number of silver atoms.
number of silver atoms.



