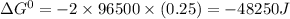11. (12pts) for the redox reaction given below, ni(s) + 2 h(aq) + ni2+(aq) + h2(e) (show your work in detail) a) write oxidation and reduction half cell reactions b) write the electrochemical cell notation c) predict whether this reaction will be spontaneous. explain. d) calculategº 6 page

Answers: 2
Other questions on the subject: Chemistry



Do you know the correct answer?
11. (12pts) for the redox reaction given below, ni(s) + 2 h(aq) + ni2+(aq) + h2(e) (show your work i...
Questions in other subjects:








Social Studies, 13.11.2019 01:31



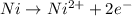
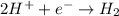
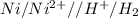
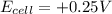 , the reaction is spontaneous.
, the reaction is spontaneous.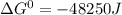
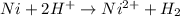
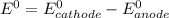
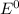 are standard reduction potentials.
are standard reduction potentials.![E^0_{[Ni^{2+}/Ni]}= -0.25V](/tpl/images/0213/4556/864ce.png)
![E^0_{[H^{+}/H_2]}=+0.0V](/tpl/images/0213/4556/826b0.png)
![E^0=E^0_{[H^{+}/H_2]}- E^0_{[Ni^{2+}/Ni]}](/tpl/images/0213/4556/3d59c.png)
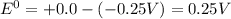
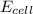 = +ve, reaction is spontaneous
= +ve, reaction is spontaneous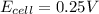 , the reaction is spontaneous.
, the reaction is spontaneous.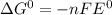
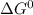 = standard gibbs free energy
= standard gibbs free energy