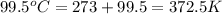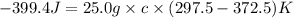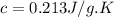A25.0g tin sample was placed in boiling water at 99.5 c until it had the same temperature. afterwards, the tin sample was placed in room temperature water. if the tin sample lost 399.4j of heat and its final temperature was 24.5 c, what is the specific heat of tin?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:20, anggar20
Harvey kept a balloon with a volume of 348 milliliters at 25.0˚c inside a freezer for a night. when he took it out, its new volume was 322 milliliters, but its pressure was the same. if the final temperature of the balloon is the same as the freezer’s, what is the temperature of the freezer? the temperature of the freezer is kelvins.
Answers: 2

Chemistry, 22.06.2019 03:40, kellypechacekoyc1b3
Chemical kinetics what was the rate of reaction in trial 3? choose the closest answer.
Answers: 3

Chemistry, 22.06.2019 19:30, xxaurorabluexx
Chlorine and water react to form hydrogen chloride and oxygen, like this: 2cl2 (g) + 2h2o (g) → 4hcl (g) + o2 (g) also, a chemist finds that at a certain temperature the equilibrium mixture of chlorine, water, hydrogen chloride, and oxygen has the following composition: compound concentration at equilibrium cl2 0.55m h2o 0.57m hcl 0.53m o2 0.34m calculate the value of the equilibrium constant kc for this reaction. round your answer to 2 significant digits.
Answers: 2
Do you know the correct answer?
A25.0g tin sample was placed in boiling water at 99.5 c until it had the same temperature. afterward...
Questions in other subjects:

Mathematics, 18.09.2020 16:01

Mathematics, 18.09.2020 16:01

Chemistry, 18.09.2020 16:01

History, 18.09.2020 16:01

Mathematics, 18.09.2020 16:01

Mathematics, 18.09.2020 16:01

Mathematics, 18.09.2020 16:01

Mathematics, 18.09.2020 16:01

Mathematics, 18.09.2020 16:01

Mathematics, 18.09.2020 16:01


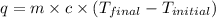
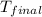 = final temperature =
= final temperature = 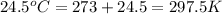
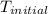 = initial temperature =
= initial temperature =