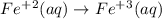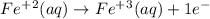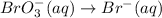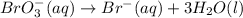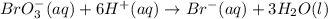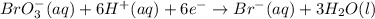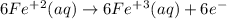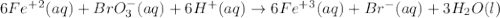Chemistry, 28.08.2019 18:20, shahfaisal
When the equation below is balanced in acidic solution using the smallest whole numbers, the coefficient in front of the h 2o( l) is (aq) bro3-(aq) → fe3 (aq) br-(aq)

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 04:30, salvadorperez26
Suppose that during that icy hot lab 65,000 j of energy were transferred to 450 g of water at 20°c what would have have been the final temperature of the water
Answers: 2

Chemistry, 22.06.2019 15:00, alanmarcus22
What does the symbol (–hfus) indicate in a phase change?
Answers: 1

Chemistry, 23.06.2019 01:30, jonmorton159
At a certain temperature the rate of this reaction is first order in hi with a rate constant of : 0.0632s2hig=h2g+i2g suppose a vessel contains hi at a concentration of 1.28m . calculate how long it takes for the concentration of hi to decrease to 17.0% of its initial value. you may assume no other reaction is important. round your answer to 2 significant digits.
Answers: 1
Do you know the correct answer?
When the equation below is balanced in acidic solution using the smallest whole numbers, the coeffic...
Questions in other subjects:







Mathematics, 02.08.2019 20:30




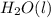 is 3.
is 3. 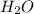 and hydrogen is balanced by adding
and hydrogen is balanced by adding 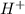 and the charge is balanced by adding electrons.
and the charge is balanced by adding electrons.