
Chemistry, 27.08.2019 22:20, aaronjin4443
Assume the solubility of benzoic acid in ice-cold water is 1.70 g/l and the solubility of benzoic acid in hot water is 68.0 g/l. calculate the minimum volume of water (in ml) needed to recrystallize 0.900 g of benzoic acid.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 21:50, BookandScienceNerd
Answer the questions about this reaction: nai(aq) + cl2(g) → nacl(aq) + i2(g) write the oxidation and reduction half-reactions: oxidation half-reaction: reduction half-reaction: based on the table of relative strengths of oxidizing and reducing agents (b-18), would these reactants form these products? write the balanced equation: answer options: a. 0/na -> +1/na+1e- b. nai(aq) + cl2(g) → nacl(aq) + i2(g) c. +1/na+1e- -> 0 /na d. -1/2i -> 0/i2+2e- e. no f. 4nai(aq) + cl2(g) → 4nacl(aq) + i2(g) g. 2nai(aq) + cl2(g) → 2nacl(aq) + i2(g) h. 4nai(aq) + 2cl2(g) → 4nacl(aq) + 2i2(g) i. nai(aq) + cl2(g) → nacl(aq) + i2(g) j. 0/cl2+2e -> -1/2cl- k. yes
Answers: 1


Chemistry, 23.06.2019 06:00, wirchakethan23
When hydrogen peroxide (h2o2) is added to potassium iodide (ki) solution, the hydrogen peroxide decomposes into water (h2o) and oxygen (o2). the chemical equation for the decomposition reaction is: 2h2o2—> 2h2o + o2. what is the role of the potassium iodide in this reaction? a. reactant. b. product. c. precipitate. d. catalyst.
Answers: 1
Do you know the correct answer?
Assume the solubility of benzoic acid in ice-cold water is 1.70 g/l and the solubility of benzoic ac...
Questions in other subjects:

History, 27.09.2019 15:30

Mathematics, 27.09.2019 15:30

Mathematics, 27.09.2019 15:30


English, 27.09.2019 15:30

Mathematics, 27.09.2019 15:30




Mathematics, 27.09.2019 15:30


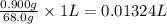 of water.
of water.



