

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, kiki197701
Agas at 155 kpa and standard temperature has an initial volume of 1.00 l. the pressure of the gas rises to 500 kpa as the temperature also rises to 135°c. what is the new volume? 2.16 l 0.463 l 0.207 l 4.82 l
Answers: 3

Chemistry, 22.06.2019 09:20, UsedForSchool2018
Which of these statements explains the difference between nuclear binding energy and the strong nuclear force ?
Answers: 3

Chemistry, 22.06.2019 17:10, hahahwha
Acalorimeter is to be calibrated: 51.203 g of water at 55.2 degree c is added to a calorimeter containing 49.783 g of water at 23.5c. after stirring and waiting for the system to equilibrate, the final temperature reached is 37.6 degree c. specific heat capacity of water (s = 4.18 j/g∙degree c). calculate the calorimeter constant. (smδt)warm water = -[(smδt)cold water + (calorimeterδtcold water)]
Answers: 2
Do you know the correct answer?
Balance the equation and show the calculation of the number of moles and grams of co2 formed from 11...
Questions in other subjects:




Mathematics, 18.07.2019 04:30

Mathematics, 18.07.2019 04:30

Arts, 18.07.2019 04:30

Mathematics, 18.07.2019 04:30


Mathematics, 18.07.2019 04:30

Mathematics, 18.07.2019 04:30


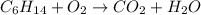
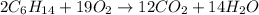
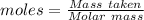
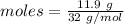
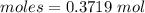
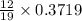 mole of carbon dioxide
mole of carbon dioxide



