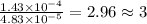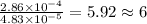Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:30, ashlpiriz123
Find the number of grams of hcl needed to react completely with .50 moles of magnesium.
Answers: 1


Chemistry, 22.06.2019 22:00, shaylasimonds587
The volume of an unknown substance in a sealed glass jar is 50 milliliters. the volume of the jar is 200 milliliters. which state of matter could the substance be?
Answers: 2

Do you know the correct answer?
The characteristic odor of pineapple is due to ethyl butyrate, a compound containing carbon, hydroge...
Questions in other subjects:




History, 04.06.2021 21:00



Chemistry, 04.06.2021 21:00


Chemistry, 04.06.2021 21:00

Mathematics, 04.06.2021 21:00




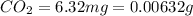
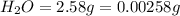
 of carbon will be contained.
of carbon will be contained. of hydrogen will be contained.
of hydrogen will be contained.