
Sulfur dioxide gas reacts with sodium hydroxide to form sodium sulfite and water. the unbalanced chemical equation for this reaction is given below: so2(g) + naoh(s) → na2so3(s) + h2o(l)assuming that you start with 36.8 g of sulfur dioxide and 20.7 g of sodium hydroxide and assuming that the reaction goes to completion, determine the mass of each product formed. g na2so3g h2o

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:30, khalaflaf2684
If 1.8 l of water is added to 2.5l of a 7.0 m koh solution, what is the molarity of the new solution
Answers: 1

Chemistry, 22.06.2019 09:40, cheesecake1919
Which diagram shows the correct way to represent an ionic compound of magnesium oxide?
Answers: 3


Do you know the correct answer?
Sulfur dioxide gas reacts with sodium hydroxide to form sodium sulfite and water. the unbalanced che...
Questions in other subjects:


English, 26.02.2021 06:30





Mathematics, 26.02.2021 06:30

Mathematics, 26.02.2021 06:30


Mathematics, 26.02.2021 06:30


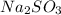 and 9.3 g of
and 9.3 g of 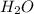 were formed
were formed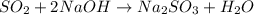
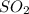 = 64 g
= 64 g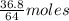 of
of 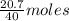 of NaOH = 0.5175 moles of NaOH
of NaOH = 0.5175 moles of NaOH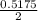 moles of
moles of 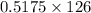 ) g = 65.2 g
) g = 65.2 g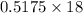 ) g = 9.3 g
) g = 9.3 g



