
Calcium reacts with bromine to form calcium bromide. 2.1 which atom will lose electrons? 2.2 how many electrons will it lose? 2.3 name the element that will gain electrons. 2.4 how many electrons does this atom gain? 2.5 write down the symbol for each of the ions formed. 2.6 write the chemical formula for calcium bromide.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:10, amuijakobp78deg
Agas mixture with a total pressure of 745 mmhg contains each of the following gases at the indicated partial pressures: co2, 245 mmhg ; ar, 119 mmhg ; and o2, 163 mmhg . the mixture also contains helium gas. part a what is the partial pressure of the helium gas? phe p h e = nothing mmhg request answer part b what mass of helium gas is present in a 10.2-l sample of this mixture at 283 k ? m m = nothing g request answer
Answers: 1


Chemistry, 23.06.2019 07:30, sweetLips230
Assignment directions: pick one of the following chemists and perform a bit of research on him/her. answer the following questions. alice hamilton rosalind franklin marie curie gertrude b. elion ada yonath henry cavendish robert boyle antoine lavoisier mario j. molina svante arrhenius
Answers: 1

Chemistry, 23.06.2019 09:00, payshencec21
How many grams of ammonia are produced when 1.0 mole of nitrogen reacts
Answers: 2
Do you know the correct answer?
Calcium reacts with bromine to form calcium bromide. 2.1 which atom will lose electrons? 2.2 how ma...
Questions in other subjects:











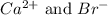
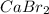
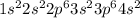
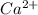 ion.
ion.![[Ar]4s^23d^{10}4p^5](/tpl/images/0191/9443/d71de.png)
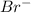 ion.
ion.



