
Chemistry, 22.08.2019 22:20, sandywarren8789
Name two compounds with more exothermic lattice energies than scandium oxide and justify your choice.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 14:40, wbrandi118
Water ionizes by the equation h2o(l)⇌h+(aq)+oh−(aq) the extent of the reaction is small in pure water and dilute aqueous solutions. this reaction creates the following relationship between [h+] and [oh−]: kw=[h+][oh−] keep in mind that, like all equilibrium constants, the value of kw changes with temperature.
Answers: 1

Chemistry, 21.06.2019 23:30, ashleyjaslin
Calculate the expected ph values of the buffer systems from the experiments (a, b,c, d), using the henderson- hasselbalch equation, ph-pka+log[a-]/[ha]. use for pka values carbonic acid= 6.37, and acetic acid= 4.75.
Answers: 2

Chemistry, 22.06.2019 03:00, ian2006huang
Which of these would be caused by a chemical change? a) the formation of lava. b) sedimantary rock layering over time. c) metamorphic rock forming from igneous. d) metamorphic rock eroding to form sedimentary rock.
Answers: 3

Chemistry, 23.06.2019 06:20, Lindsay882
Type the correct answer in each box. balance the chemical equation.__ n203 ➡️ __ n2 +__ o2
Answers: 1
Do you know the correct answer?
Name two compounds with more exothermic lattice energies than scandium oxide and justify your choice...
Questions in other subjects:

Physics, 23.10.2020 02:01

Mathematics, 23.10.2020 02:01

English, 23.10.2020 02:01


Mathematics, 23.10.2020 02:01

Mathematics, 23.10.2020 02:01

Spanish, 23.10.2020 02:01

Computers and Technology, 23.10.2020 02:01

Physics, 23.10.2020 02:01

Mathematics, 23.10.2020 02:01


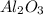
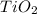
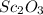 ) is an oxide in which
) is an oxide in which 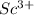 behaves as cation and
behaves as cation and 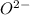 behaves as anion.
behaves as anion.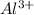 is smaller than
is smaller than 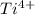 is greater than that of
is greater than that of 



