Chemistry, 22.08.2019 17:10, zoeyandblaze
Write the expression for the equilibrium constant kp for the following reaction. enclose pressures in parentheses and do not write the chemical formula as a subscript. for example, enter (pnh3)2 as (p nh3)2 .if either the numerator or denominator is 1, enter 1nh4hs(s) ↔ nh3(g) + h2s(g)

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:30, giraffegurl
If 34.2 grams of lithium react with excess water, how many liters of hydrogen gas can be produced at 299 kelvin and 1.21 atmospheres? 2 li (s) + 2 h2o (l) yields 2 lioh (aq) + h2 (g)
Answers: 3

Chemistry, 22.06.2019 10:00, micahwilkerson9495
Select all of the methods through which a drug can enter your body. injection swallowing inhalation absorption
Answers: 2


Chemistry, 22.06.2019 17:40, adantrujillo1234
Areaction in which products can react to re-form reactants is
Answers: 1
Do you know the correct answer?
Write the expression for the equilibrium constant kp for the following reaction. enclose pressures i...
Questions in other subjects:



Mathematics, 06.07.2019 06:00


Geography, 06.07.2019 06:00

History, 06.07.2019 06:00

Spanish, 06.07.2019 06:00

Mathematics, 06.07.2019 06:00


English, 06.07.2019 06:00


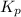 is written below.
is written below.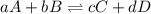
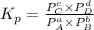
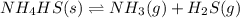
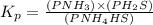
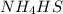 will be 1 because it is solid.
will be 1 because it is solid.