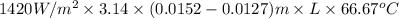Aheat exchanger is to be constructed by forming copper tubing into a coil and placing the latter inside an insulated steel shell. in this exchanger, water will flow inside the tubing, and a hydrocarbon vapor at a rate of 0.126 kg/s will be condensing on the outside surface of the tubing. the inside and outside diameters of the tube are 0.0127 and 0.0152 m, respectively, inlet and exit temperatures for the water are 10 and 32°c, respectively. the heat of conden- sation of the hydrocarbon at a condensing temperature of 88°c is 335 kj/kg, and the heat- transfer coefficient for the condensing vapor is 1420 w/m².k. heat losses from the shell may be neglected. what length of copper tubing will be required to accomplish the desired heat transfer?

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 04:00, kichensides
Asample of aluminum foil contains 8.60 × 1023 atoms. what is the mass of the foil?
Answers: 1

Chemistry, 22.06.2019 06:00, jwood287375
When a spring is compressed, the energy changes from kinetic to potential. which best describes what is causing this change?
Answers: 3

Chemistry, 22.06.2019 16:10, 00015746
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2
Do you know the correct answer?
Aheat exchanger is to be constructed by forming copper tubing into a coil and placing the latter ins...
Questions in other subjects:


Mathematics, 27.04.2021 02:30

Mathematics, 27.04.2021 02:30




Chemistry, 27.04.2021 02:30




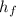 ) = 335 kJ/kg
) = 335 kJ/kg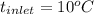 , and
, and 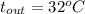
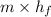
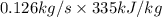
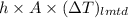
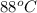 .
.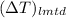 =
= 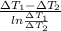
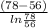
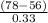
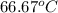
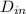 = 0.0127, and
= 0.0127, and 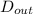 = 0.0152 m
= 0.0152 m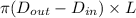 ......... (1)
......... (1)