
Chemistry, 08.01.2020 20:31, oofoofoof1
An atom of radon gas emits an alpha particle as shown below. radon-222 has 86 protons and 136 neutrons. what daughter product will form from this decay? francium-222 (87 protons) polonium-218 (84 protons) radium-226 (88 protons) lead-220 (82 protons)

Answers: 1
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 21:30, sierradanielle9280
In science class richard learns that a substance has a boiling point of 230 fahrenheit his teacher ask him to convert this temperature to degrees celsius what is the boiling point of his substance in degrees celsius
Answers: 3

Chemistry, 23.06.2019 06:40, hackman1216
The combustion of methane, ch4, releases 890.4kj/mol. that is, when one mole of methane is burned,890.4 kj are given off to the surroundings. this meansthat the products have 890.4 kj less than the reactants. thus, ah for the reaction = - 890.4 kj. a negative symbolforah indicates an exothermic reaction. ch (g) + 20 (g)> co2 (g) + 2 h0 (1); ah = - 890.4 kga) how much energy is given off when 2.00 mol of ch, are burned? b) how much energy is released when 22.4g of ch. areburned?
Answers: 1
Do you know the correct answer?
An atom of radon gas emits an alpha particle as shown below. radon-222 has 86 protons and 136 neutro...
Questions in other subjects:

Chemistry, 12.06.2020 23:57


History, 12.06.2020 23:57


Physics, 12.06.2020 23:57


History, 12.06.2020 23:57




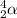 .
.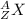 →
→ 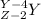 +
+ 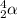 .
.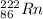 →
→ 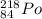 +
+ 



