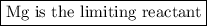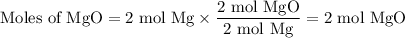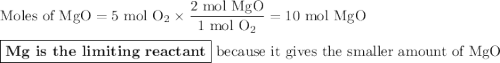Chemistry, 20.08.2019 04:20, KevinReed6444
Two moles of magnesium (mg) and five moles of oxygen (o2) are placed in a reaction vessel. when magnesium is ignited, it reacts with oxygen. what is the limiting reactant in this experiment?
mg + o2 → mgo (unbalanced)
a.
mg
b.
o2
c.
mgo
d.
both the reactants are in same proportion

Answers: 1
Similar questions


Chemistry, 28.08.2019 07:30, lulustar13
Answers: 1


Chemistry, 16.10.2019 04:30, honey66
Answers: 1
Do you know the correct answer?
Two moles of magnesium (mg) and five moles of oxygen (o2) are placed in a reaction vessel. when magn...
Questions in other subjects:










Biology, 20.01.2021 01:30