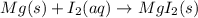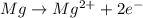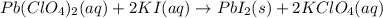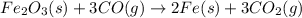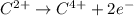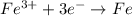Chemistry, 16.08.2019 08:10, avastanleyy
Which of the following chemical equations show(s) oxidation-reduction reactions? 1. mg(s) + i2(aq) --> mgi2(s)2. pb(clo4)2(aq) + 2 ki(aq) --> pbi2(s) + 2 kclo4(aq)3. fe2o3(s) + 3 co(g) --> 2 fe(s) + 3 co2(g)1 only2 only1 and 21 and 32 and 3

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:30, mandy9386
You are making a solution of calcium chloride dissolved in water. you add solid, stir, and it dissolves. you add just a spatula tip full, stir, and the solid does not dissolve. how could you describe the solutions before and after adding the spatula tip amount
Answers: 1

Chemistry, 22.06.2019 17:10, glitterpanda2468
Calculate the estimated density of each ball. use the formula d = m/v where d is the density, m is the mass, and v is the volume. record your calculations in table a of your student guide. given that the density of water is 1.0 g/cm3, make a prediction about whether each ball will float in water. record your prediction in table a. what is the estimated density of the table tennis ball? record your answer to the nearest hundredth
Answers: 2

Chemistry, 23.06.2019 06:00, tytianadyson74
What volume of argon gas is equal to 1.60 grams of argon
Answers: 1

Chemistry, 23.06.2019 06:30, Liapis
An engineer decides to use a slightly weaker material rather than a stronger material, since she knows that the stronger material can break suddenly. this is an example of what? a choosing a material that will show warning before it fails b using composite materials that combine strength c using a material for multiple applications d using design techniques that increase efficiency and reduce cost
Answers: 3
Do you know the correct answer?
Which of the following chemical equations show(s) oxidation-reduction reactions? 1. mg(s) + i2(aq) -...
Questions in other subjects:

Mathematics, 06.10.2021 22:10






Computers and Technology, 06.10.2021 22:10

Social Studies, 06.10.2021 22:10


Biology, 06.10.2021 22:10