
Apossible mechanism for the gas phase reaction of no and h2 is as follows: step 1 2no n2o2 step 2 n2o2 + h2 n2o + h2o step 3 n2o + h2 n2 + h2o which of the following statements concerning this mechanism is not directly supported by the information provided? a. n2o2 is an intermediate. b. all steps are bimolecular reactions. c. the rate expression for step 1 is rate = k[no]2. d. step 1 is the rate determining step. e. there is no catalyst in this reaction. 1 points question 9

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 17:10, glitterpanda2468
Calculate the estimated density of each ball. use the formula d = m/v where d is the density, m is the mass, and v is the volume. record your calculations in table a of your student guide. given that the density of water is 1.0 g/cm3, make a prediction about whether each ball will float in water. record your prediction in table a. what is the estimated density of the table tennis ball? record your answer to the nearest hundredth
Answers: 2

Chemistry, 22.06.2019 20:10, jakhunter354
The lattice enthalpy (formation of ionic solid from ions in the gas phase) for agcl(s) is -916 kj/mol and the hydration enthalpy (dissolution of gaseous ions into water) is -850 kj/mol. how much heat (in joules) is involved in forming 1l of saturated agcl solution (1.8 × 10-4 g / 100 ml water) by dissolving agcl(s)? assume solution volume does not change much upon dissolution. the equations are given below. ag+(g) + cl−(g) æ agcl(s)
Answers: 3

Chemistry, 22.06.2019 23:30, bxymichelle
With the largest atoms and the smallest number of valence electrons and with the smallest atoms and the greatest number of valence electrons are the most reactive. a. nonmetals; metals b. nonmetals; transition elements c. transition elements; metals d. metals; nonmetals
Answers: 3
Do you know the correct answer?
Apossible mechanism for the gas phase reaction of no and h2 is as follows: step 1 2no n2o2 step 2 n...
Questions in other subjects:

Chemistry, 25.10.2021 07:30

English, 25.10.2021 07:30

History, 25.10.2021 07:30


Biology, 25.10.2021 07:30

Medicine, 25.10.2021 07:30


Physics, 25.10.2021 07:30

Biology, 25.10.2021 07:30


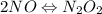 (fast)
(fast)![[NO]^{2}](/tpl/images/0174/7336/f0d3d.png)
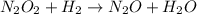
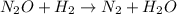 (fast)
(fast)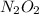 is intermediate in nature.
is intermediate in nature.



