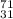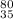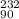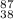Chemistry, 12.08.2019 19:10, jackiecroce1
Write the symbol for each of the following ions:
(a) the ion with a 3+ charge, 28 electrons, and a mass number of 71
(b) the ion with 36 electrons, 35 protons, and 45 neutrons
(c) the ion with 86 electrons,142 neutrons, and a 4+ charge
(d) the ion with a 2+ charge, atomic number 38, and mass number 87

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:50, mrylenastewart
What are transitions between a liquid and a solid called? identify which way they are transitioning
Answers: 2


Do you know the correct answer?
Write the symbol for each of the following ions:
(a) the ion with a 3+ charge, 28 electrons,...
(a) the ion with a 3+ charge, 28 electrons,...
Questions in other subjects:

Computers and Technology, 06.12.2019 20:31

English, 06.12.2019 20:31

Biology, 06.12.2019 20:31

English, 06.12.2019 20:31


Computers and Technology, 06.12.2019 20:31

Mathematics, 06.12.2019 20:31



Physics, 06.12.2019 20:31