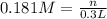Chemistry, 08.08.2019 06:20, jsharma57p7enrw
In the laboratory, a student adds 19.7 g of barium acetate to a 500. ml volumetric flask and adds water to the mark on the neck of the flask. calculate the concentration (in mol/l) of barium acetate, the barium ion and the acetate ion in the solution. [ba(ch3coo)2] = m [ba2+] = m [ch3coo-] = m
calculate the mass, in grams, of iron(ii) sulfate that must be added to a 300-ml volumetric flask in order to prepare 300 ml of a 0.181 m aqueous solution of the salt.
grams

Answers: 2
Other questions on the subject: Chemistry



Do you know the correct answer?
In the laboratory, a student adds 19.7 g of barium acetate to a 500. ml volumetric flask and adds wa...
Questions in other subjects:











![[Ba(CH_3COO)_2]=0.1545 mol/L](/tpl/images/0173/1748/733d1.png)
![[Ba^{2+}]=0.1545 mol/L](/tpl/images/0173/1748/704db.png)
![[CH_3COO^-]=0.3090 mol/L](/tpl/images/0173/1748/1e1ae.png)
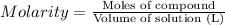
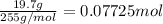
![[Ba(CH_3COO)_2]=\frac{0.07725 mol}{0.5L}=0.1545 mol/L](/tpl/images/0173/1748/ea20e.png)
![[Ba^{2+}]=1\times [Ba(CH_3COO)_2]](/tpl/images/0173/1748/fd1e3.png)
![[Ba^{2+}]=1\times 0.1545 mol/L=0.1545 mol/L](/tpl/images/0173/1748/1f46a.png)
![[CH_3COO^-]=2\times [Ba(CH_3COO)_2]](/tpl/images/0173/1748/6fb75.png)
![[CH_3COO^-]=2\times 0.1545 mol/l=0.3090 mol/L](/tpl/images/0173/1748/e3fff.png)
![[Fe_2(SO_4)_3]=0.181 M](/tpl/images/0173/1748/b830a.png)