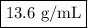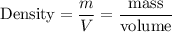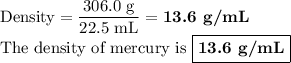Chemistry, 08.08.2019 00:30, CamBroOkaii2590
Mercury metal is poured into a graduated cylinder that holds exactly 22.5 ml. the mercury used to fill the cylinder weighs 306.0 grams. from this information, calculate the density of the mercury.

Answers: 3
Other questions on the subject: Chemistry



Do you know the correct answer?
Mercury metal is poured into a graduated cylinder that holds exactly 22.5 ml. the mercury used to fi...
Questions in other subjects:




History, 17.06.2021 17:40


Mathematics, 17.06.2021 17:40

Engineering, 17.06.2021 17:40

Arts, 17.06.2021 17:40