
Chemistry, 06.08.2019 00:30, tybreyonnaHco7855
In the haber process for ammonia synthesis, k " 0.036 for n 2 (g) ! 3 h 2 (g) ∆ 2 nh 3 (g) at 500. k. if a 2.0-l reactor is charged with 1.42 bar of n 2 and 2.87 bar of h 2 , what will the equilibrium partial pressures in the mixture be?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 23.06.2019 03:00, amberskids2
You have a sample of a metal, the sample is exactly 6.02 x 1023atom, if the sample has a mass 55.85 what metal is your sample made of?
Answers: 2



Chemistry, 23.06.2019 09:00, msladycie8831
Avogradoa number was calculated by determining the number of atoms in?
Answers: 1
Do you know the correct answer?
In the haber process for ammonia synthesis, k " 0.036 for n 2 (g) ! 3 h 2 (g) ∆ 2 nh 3 (g) at 500....
Questions in other subjects:



Mathematics, 24.11.2019 07:31


Biology, 24.11.2019 07:31



Biology, 24.11.2019 07:31

English, 24.11.2019 07:31


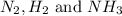 at equilibrium are, 1.133, 2.009, 0.574 bar respectively. The total pressure at equilibrium is, 3.716 bar
at equilibrium are, 1.133, 2.009, 0.574 bar respectively. The total pressure at equilibrium is, 3.716 bar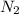 = 1.42 bar
= 1.42 bar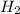 = 2.87 bar
= 2.87 bar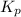 = 0.036
= 0.036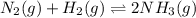
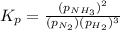
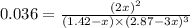
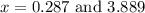
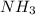 at equilibrium = 2x = 2 × 0.287 = 0.574 bar
at equilibrium = 2x = 2 × 0.287 = 0.574 bar




