
Chemistry, 30.07.2019 22:20, palcochran1313
Given the following balanced reaction between liquid ammonia and oxygen gas to produce nitrous oxide gas and water, how many grams of water, h2o, are produced from 317 grams of ammonia and excess oxygen? (to find the molar mass in the problem use the periodic table and round the mass to the hundreds place for calculation).
(a) 224 g
(b) 335 g
(c) 503 g

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 19:50, VoidedAngel
When the mercury level in a barometer decreases that atmospheric pressure has
Answers: 3


Chemistry, 22.06.2019 23:00, catdog5225
What is formed when amino acids form long chains or polymerize
Answers: 1

Chemistry, 22.06.2019 23:50, datboyjulio21
Which scientists contributed to the determination of how cfcs in clouds in the upper atmosphere could destroy ozone molecules
Answers: 1
Do you know the correct answer?
Given the following balanced reaction between liquid ammonia and oxygen gas to produce nitrous oxide...
Questions in other subjects:

Mathematics, 23.06.2020 19:01


Mathematics, 23.06.2020 19:01

Mathematics, 23.06.2020 19:01

Mathematics, 23.06.2020 19:01



Mathematics, 23.06.2020 19:01



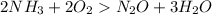


 to moles
to moles 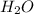 by using mole ratio of
by using mole ratio of 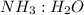 i.e., 2 : 3
i.e., 2 : 3

 is formed.
is formed.
 is
is 





